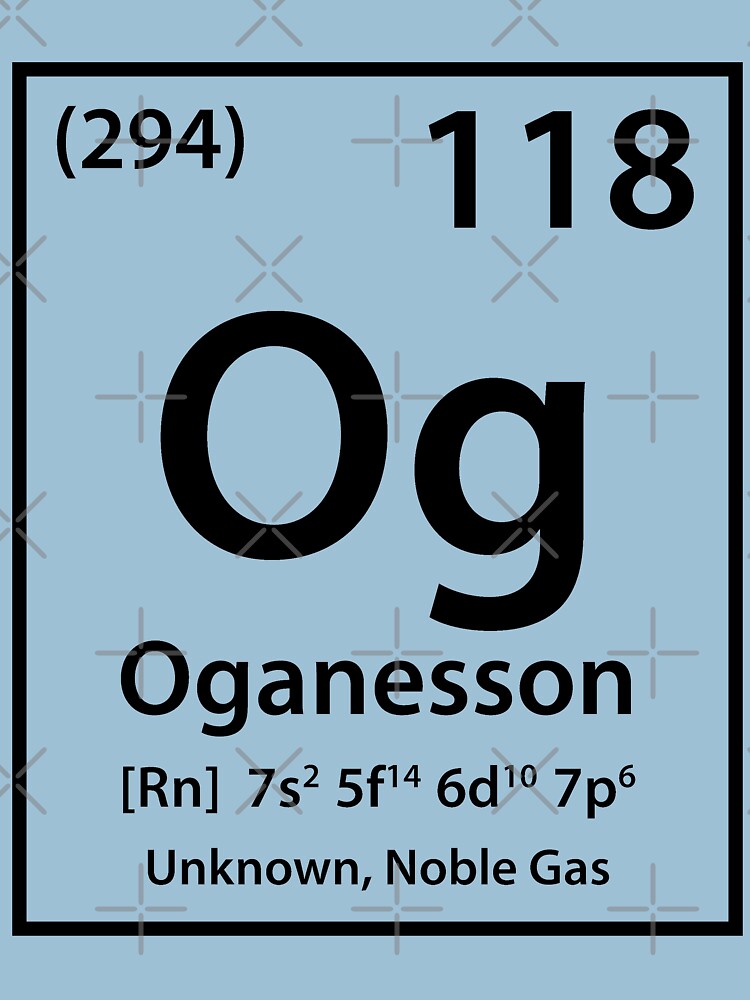
One would predict its behaviour to be similar to that of radon (immediately above oganesson in the periodic table) and xenon (two places above). Reaction of oganesson with basesĪs no atoms of oganesson have ever been made, its reactivity with bases is unknown. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. One would predict its behaviour to be similar to that of radon (immediately above oganesson in the periodic table) and xenon (two places above). Oganesson is a chemical element with atomic number 118 which means there are 118 protons and 118 electrons in the atomic structure. Reaction of oganesson with acidsĪs no atoms of oganesson have ever been made, its reactivity with acids is unknown. Reaction of oganesson with the halogensĪs no atoms of oganesson have ever been made, its reactivity with the halogens is unknown. It would probably dissolve to some extent in water at 20☌ (293 K). 
However, based upon the behaviour of radon (immediately above oganesson in the periodic table), one would predict that it does not react with water. Reaction of oganesson with waterĪs no atoms of oganesson have ever been made, its reactivity with water is unknown. However, based upon the behaviour of radon (immediately above oganesson in the periodic table), one would predict that it does not react with air. Although the element is positioned in the noble gas column (group 18) of the periodic table, theorists believe that it probably is neither a gas nor “noble”-that is, unreactive.As no atoms of oganesson have ever been made, its reactivity with air is unknown. Oganesson-294’s extremely short half-life precludes measurements of its physical and chemical properties. Computer modeling shows that more stable isotopes of oganesson may have atomic masses as high as 313. In 2011, researchers at the GSI Helmholtz Center for Heavy Ion Research (Darmstadt, Germany) reported the possible discovery of 295Og, but this finding has not been confirmed. At that time, Oganessian and colleagues reported an 294Og half-life of 0.69 ms. In 2015, the Joint Working Party of the International Union of Pure and Applied Chemistry and the International Union of Pure and Applied Physics recognized the discovery of oganesson. Additional experiments to confirm the existence of 249Og continued for the next decade. The nuclear reaction was later established to be 249Cf + 48Ca → 294Og + 3n, where n stands for neutron. They bombarded atoms of californium-249 with calcium-48 atoms to produce a few nuclei with atomic mass 294. The team’s first indication that it had produced element 118 came in 2002. The team consists of scientists at the Joint Institute for Nuclear Research (Dubna, Russia) and Lawrence Livermore National Laboratory (CA). Oganesson is named after Russian nuclear physicist Yuri Oganessian, a leader of the Russian–American team that created most of the elements with atomic numbers >110. This produced oganesson-294, an isotope with a half-life of about 0.89 milliseconds (0.00089 seconds), and three free neutrons. This composition has not been confirmed experimentally. This is something of a “magic” number because it completes the seventh row of the periodic table of elements. 
To date, the highest atomic number element to be discovered or synthesized is oganesson (symbol Og), with atomic number 118. We take “last element” to mean the element with the highest atomic number. 
Even though this would be an atom and not a molecule, we thought it would be of interest to our readers. Last month, a junior high school student in New Jersey suggested that we feature “a known isotope of the last element of the periodic table”.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
